Soil Sample 5508 / Culture 10520 / Isolate 10521
Isolate 10521
MGKE1 University of Connecticut
Published
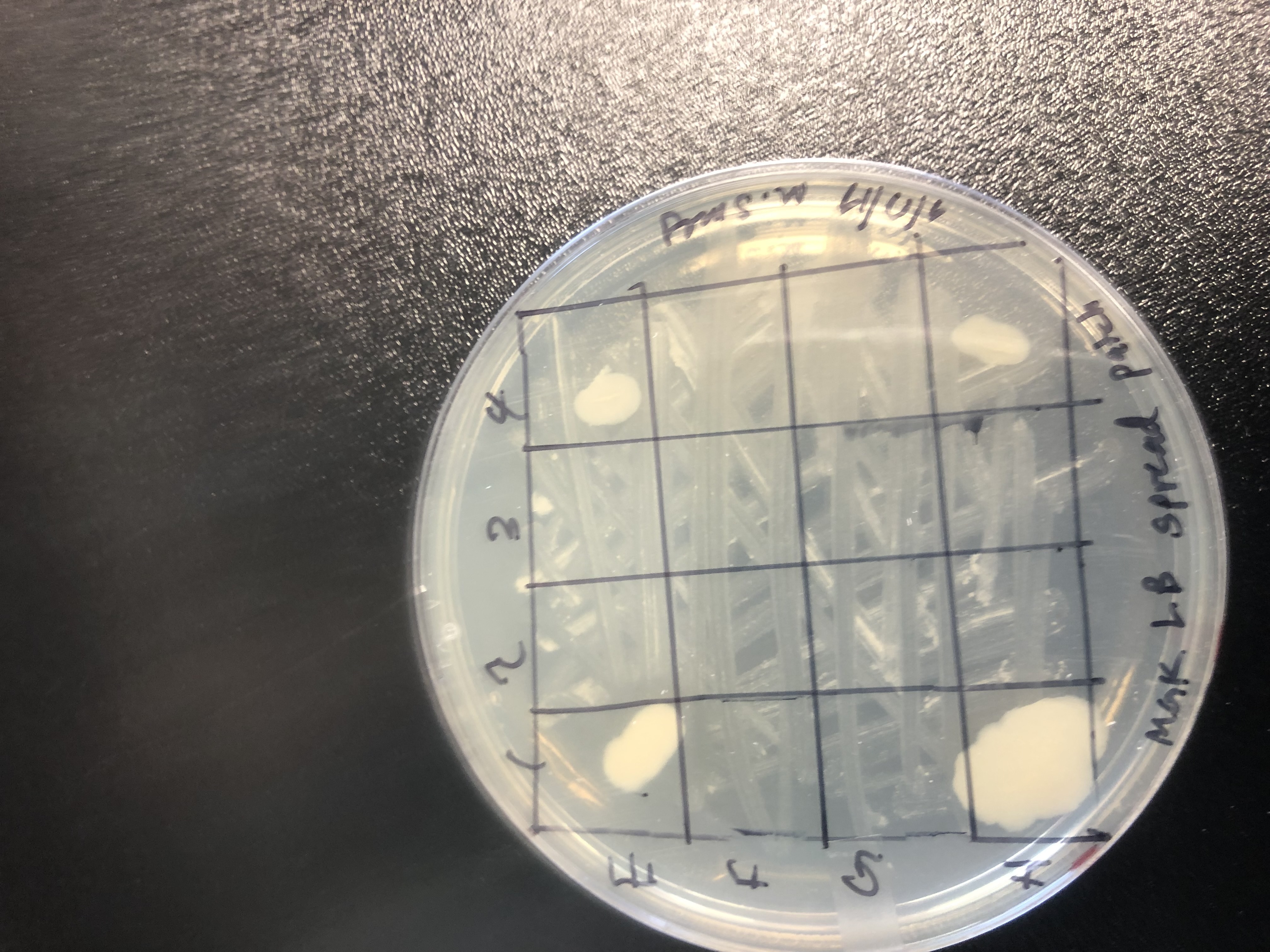
ESKAPE Test
Published
Recorded Friday, December 6, 2019
| Tested against ESKAPE relative | Antibiotic activity | Length of incubation | Media used |
|---|---|---|---|
| Bacillus subtilis | No | ||
| Enterococcus raffinosus | No | ||
| Staphylococcus epidermidis | Yes | 24.0 hr | LB |
| Escherichia coli | No | ||
| Acinetobacter baylyi | No | ||
| Pseudomonas putida | No | ||
| Enterbacter aerogenes | No | ||
| Mycobacterium smegmatis | Yes | 24.0 hr | LB |
16S rRNA Test
Published
Recorded Friday, December 6, 2019
Genus
unknownFASTA sequence
MGKG1_r-1391R.seq
DOWNLOAD FILE
MGKG1_f-27F.seq
DOWNLOAD FILE
Distinguishing Characteristics
87% match to unculturable bacteria found in apple tree rootsAntibiotic Activity of Extract
Published
Recorded Friday, December 6, 2019
Solvent used
MethanolExperiment Description
Metabolite extraction -obtain Swab plates of isolates -using a sterile spatula cut the agar into 1mm cubes -place cut up agar into screw-top jar and label with respective isolate names -place screw top jar containing agar into cooler with a dry ice and ethanol bath -allow a deep freeze of agar for at least 20 minutes -remove screw top jar from dry ice and ethanol bath under a hood add 15ml of ethyl acetate and 10ml of distilled water to jar. -allow mixture to incubate on a shaker for 24 hours -obtain two scintillation vials for each isolate -label one of the vials for each isolate extract and measure and record the mass of the empty vial -remove mixture in screw top jars from incubation -using a sterile transfer pipette transfer all liquid in the screw top jar to the unlabeled scintillation vial -allow liquid to settle in scintillation jar -using another sterile pipette transfer the top layer of settled solution into the labeled scintillation vial -let vial sit opened in hood to dry down over a 3-4 day period -After solution has dried down measure and record the mass of the scintillation vial -using the difference in mass between the empty scintillation vial and scintillation vial with extract add methanol to get a concentration of 10 micrograms per milliliter. -use an ultrasonic cleaner in order to homogenize mixture Agar overlay -obtain and label LB plates for each ESKAPE safe relative isolates showed antibiotic activity against -draw labeled circles on bottom of plate for each extract -pipette 10 microliters of extract into labeled circle and allow to dry -repeat above step two more times for this extract -place drops of all other isolates following steps detailed above ensuring to use a sterile pipette tip for each isolate and include a methanol control on each plate as well. -after extracts have dried into agar obtain a test-tube of liquid agar warmed in a heated water bath -add 100-200 microliters of one of the ESKAPE safe relatives to be tested in liquid media to the test-tube containing liquid agar -spin test-tube with inoculated agar between hands to ensure even distribution of bacteria -pour liquid agar over labeled LB plate with extract dried into agar and allow agar to completely dry and become solid -repeat above steps for all ESKAPE safe relatives to be tested on each of the extract containing LB plates prepared ensuring to use a fresh test-tube of liquid agar for each ESKAPE safe relative to be tested -incubate plates at 27 degrees Celsius until next lab period -remove plates from incubation and observe for zones of inhibition -record results| Tested against ESKAPE relative | Antibiotic activity | Length of incubation | Media used |
|---|---|---|---|
| Bacillus subtilis | No | ||
| Enterococcus raffinosus | No | ||
| Staphylococcus epidermidis | Yes | 24.0 hr | LB |
| Escherichia coli | No | ||
| Acinetobacter baylyi | No | ||
| Pseudomonas putida | No | ||
| Enterbacter aerogenes | No | ||
| Mycobacterium smegmatis | Yes | 24.0 hr | LB |
Eukaryotic Inhibition
Published
Recorded Friday, December 6, 2019
Experiment description
Metabolite extraction -obtain Swab plates of isolates -using a sterile spatula cut the agar into 1mm cubes -place cut up agar into screw-top jar and label with respective isolate names -place screw top jar containing agar into cooler with a dry ice and ethanol bath -allow a deep freeze of agar for at least 20 minutes -remove screw top jar from dry ice and ethanol bath under a hood add 15ml of ethyl acetate and 10ml of distilled water to jar. -allow mixture to incubate on a shaker for 24 hours -obtain two scintillation vials for each isolate -label one of the vials for each isolate extract and measure and record the mass of the empty vial -remove mixture in screw top jars from incubation -using a sterile transfer pipette transfer all liquid in the screw top jar to the unlabeled scintillation vial -allow liquid to settle in scintillation jar -using another sterile pipette transfer the top layer of settled solution into the labeled scintillation vial -let vial sit opened in hood to dry down over a 3-4 day period -After solution has dried down measure and record the mass of the scintillation vial -Add 1ml of methanol to scintillation vial and record the concentration -use an ultrasonic cleaner in order to homogenize mixture -perform dilutions as necessary to get concentrations of 10mg/ml, 25mg/ml and 50mg/ml Chia seeds -Obtain and label water agar plates for each of the isolate extracts at concentrations of 10mg/ml, 25mg/ml and 50mg/ml to be tested positive control using tetracycline at a concentration of 5mg/ml. And negative control of methanol -Draw circles on the bottom of the plates for each treatment -using forceps place 5 chia seeds in each circle -Pipette 10 microliters of each treatment into their respective labeled circles. -Repeat above step three times for each extract allowing it to dry in between -Incubate water agar plates at room temperature with adequate sunlight| Eukaryotic organism | Eukaryotic inhibition |
|---|---|
| chia seeds | No |
Associated Entries
Soil Sample 5508
Collected Thursday August 29, 2019
Published
Culture Conditions 10520
Recorded Friday December 6, 2019
Published
Isolate 10521
Recorded Friday December 6, 2019
Published
Culture Conditions 10518
Recorded Friday December 6, 2019
Published
Isolate 10519
Recorded Friday December 6, 2019
Published